
This study aimed to determine whether aromatherapy inhalation would reduce chemotherapy-induced nausea and vomiting (CINV) among cancer patients receiving moderate to high emetogenic chemotherapy (HEC) regimen.
AUTHORS
Debbie Anglade, Caterine Diaz, Roberto Roman Laporte, Karina Gattamorta,
Sameena F. Sheikh-Wu, Yhenifer Diaz Granados, Hoyan Ng-Chen, Junet Alvarez,
Lisa F. Rosen, Joseph Pizzolato
DATE: August 13 2025
BACKGROUND
Despite numerous antiemetic agents and evidence-based guidelines, chemotherapy-induced nausea and vomiting (CINV) impacts approximately 80 % of cancer patients undergoing chemotherapy (Ahn et al., 2024), and nearly 40 % of cancer patients receiving chemotherapy are not properly treated (Dranitsaris et al., 2017; Gupta et al., 2021; Yokoe et al., 2018; Mapp et al., 2020). Even with medical and treatment advancements, CINV remains a significant clinical challenge for patients undergoing chemotherapy (American Cancer Society, 2020; Escobar et al., 2015). CINV negatively impacts quality of life (QOL) (Dranitsaris et al., 2017; Mosa et al., 2020; Barbour, 2017) and is a feared adverse event associated with significant morbidity (Escobar et al., 2015; Ng et al., 2015). Therefore, CINV contributes to non-compliance and unwillingness to continue with chemotherapy treatment (Dranitsaris et al., 2017; Gupta et al., 2021).
Another consideration is the potential emetic risk of the individual chemotherapy agents, including frequency and severity of CINV. Low emetic risk chemotherapy regimens often require a single antiemetic agent, while higher emetic risk chemotherapy (HEC) regimens call for a multimodal approach involving a combination of several different antiemetic agents (Cao et al., 2024). Antiemetic agents are shown to be effective in managing CINV; however, they can have significant negative effects such as drug-drug interactions, adverse drug reactions and unpleasant side effects. Balancing the benefits and risks is essential in providing safe and optimal care (Adel, 2017).
A recent study showed that the use of aromatherapy, in addition to antiemetic therapy, can reduce CINV during the acute phase of chemotherapy (Ahn et al., 2024; Farahani et al., 2019). Additionally, aromatherapy is recommended as a complementary treatment forpostoperative nausea and vomiting due to its safe and rapid onset, ease of administration, minimal drug interaction, and benefits that have been shown to reduce anxiety and depression, and increase patients’ well-being (Duff, 2009; Asay et al., 2018).
The present study examined the efficacy of a nurse-led aromatherapy inhalation intervention as a non-pharmacological approach to managing CINV among outpatients receiving moderate-to HEC regimens. The primary objective was to evaluate the effect of aromatherapy inhalationon the severity and frequency of CINV. Secondary objectives included assessing its impact on antiemetic medication use, psychological distress (stress, anxiety, and depression), resilience, and overall QOL. It was hypothesizedthat participants receiving aromatherapy inhalation would experience a significant reduction in CINV severity, decreased reliance on antiemetic medications, lower levels of psychological distress, and improved resilience and QOL compared with those not receiving the intervention.
METHODS
Study design and sample size
One hundred participants receiving moderate to HEC treatment were enrolled in a two arm control trial design (NCT05319860) at a National Cancer Institute-Designated cancer center’s comprehensive treatment unit (CTU). The Food and Drug Administration (FDA) and the Institutional Review Board (IRB) approved the study prior to data collection. Participants signed electronic informed consent and HIPAA waiver forms prior to data collection. The study recruitment took place at an academic medical center in the southeastern region of the United States. Data collection occurred between August 2022 and April 2024.
Study procedures
Participants were identified through a weekly review of the CTU schedule patient notes, which display patients’ chemotherapy regimen summary (e.g., chemotherapy drug type, dosage, and frequency). Patients identified undergoing moderate to HEC (adjuvant or neoadjuvant) were then approached during their regular outpatient CTU visit. A study team member explained the study to the potential participant, if eligible, and the potential participant agreed, informed consent was then obtained. The consented participants were
sequentially randomized (Urbaniak and Plous, 2013) to the control or the intervention groupusing a pre-generated random allocation sequence created through a computerized random number generator (simple randomization) maintained in a password-protected electronic spreadsheet available to the principal investigator (PI) and co-PI only. Participants were assigned to groups in the order of their enrollment according to the concealed sequence. To ensure adherence and create an auditable trail, the PI/co-PI were mandated to assign the participant the next sequential number on the randomization list immediately after consent was signed by the participant. This assignment (Participant ID and Group) was then immediately recorded in the REDCap database by the study team, and the PI/co-PI verified that the predetermined sequential order was followed, reducing the opportunity for the research team to deviate from the randomization assignment.
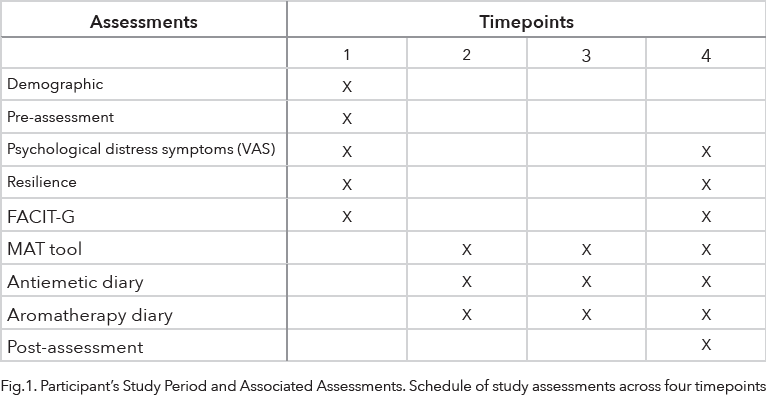
Consented study participants were seen by a study team member over four timepoints throughout the treatment, during which the participants completed the study instruments (Fig. 1). Participants in the control group followed physician-prescribed antiemetic treatment. Participants in the intervention group received an aromatherapy inhaler and were instructed to use the inhaler at the first signs of CINV and as needed. If no relief was obtained, they were to follow their physician’s prescribed antiemetic treatment.
Inclusion and exclusion criteria
Inclusion criteria were a) 18 years of age or older; b) able to read and speak English or Spanish; c) able to and willing to give informed consent; d) currently undergoing only moderate to HEC (adjuvant or neoadjuvant) treatment; e) have three or more remaining cycles of chemotherapy; and f) symptoms of nausea or vomiting after their first chemotherapy infusion. Exclusion criteria included a) unable or unwilling to give informed consent; b) sensitivity to essential oils; c) olfactory disorders; d) hormone-sensitive cancer diagnosis; e) receiving chemotherapy for the first time; and f) undergoing low emetogenic chemotherapy regimens.
Intervention
The aromatherapy inhaler intervention is used at the first sign of CINV and subsequently as needed. The aromatherapy inhaler contains a proprietary blend of peppermint, spearmint, lavender, and ginger essential oils (QueaseEASE, Soothing Scents, Newton, AL); product details can be found in the supplement information. To reduce variability in the intervention delivery, this standardized, commercially available aromatherapy inhaler product was utilized. Using a premanufactured product ensured consistency in essential oil concentration, formulation, and delivery mechanism across all participants. This approach minimized potential bias associated with product preparation or dosing variability and enhanced the reproducibility of the intervention. Participants used the inhaler by twisting open the inhaler cap, holding the inhaler below their nose, and taking deep breaths to activate the aromatherapy. After inhalation, the participant replaced the cap on the device. The aromatherapy inhaler can be used as needed, with unlimited frequency, for up to six months. There are no known contraindications to the use of the inhaler.
Outcome measurements
The study used established instruments with scoring procedures and validity evidence. The data measures were collected in REDCap, an electronic database (Harris et al., 2019).
The researcher created instruments, including an 18-item self-report Pre-Assessment Demographic Checklist that collected demographic information and risk factors for CINV. The pre-assessment demographic checklist provided the descriptive characteristics of the study’s sample population. Additional data, such as medical history, family history, and recent outpatient and inpatient clinic or hospital visits, were extracted from the electronic health record.
The Multinational Association of Supportive Care Antiemesis Tool (MAT) is designed to measure acute and delayed CINV across patients’ entire chemotherapy regimen and is used to facilitate discussions about CINV with healthcare providers (Ahmad, 2016; Molassiotis et al., 2007a,b; Yamaguchi et al., 2009). The tool is available in English and Spanish and consists of eight items that assess participants’ CINV. The first four items assess the participants’ acute emesis phase, and the last four items assess the delayed emesis phase.
The study team created and provided an Aromatherapy Diary for the participants to record their weekly usage by date and time, as well as an evaluation of the aromatherapy. The evaluation assessed the effects of aromatherapy on CINV by asking the participants, “Did the aromatherapy take away your nausea?” Answers ranged from “it took away my nausea completely” to “it had no effect on my nausea.” If the participants did not get relief from the aromatherapy, the participants had the option to take the prescribed antiemetic.
If antiemetic medication was used, participants documented the medication in the aromatherapy diary by marking the option titled “med.”
The participants also recorded the use of medication or complementary methods as needed in an Antiemetic Diary. The study team created this diary for the participants to record any antiemetic medication and complementary methods used by date, time, and type/name of antiemetic medications or complementary treatment, along with a description of relief from CINV.
The Connor-Davidson Resilience Scale (CD-RISC) assesses participants’ resilience pre and post-intervention (Burns and Anstey, 2010). For this study, we used the 10-item questionnaire that includes questions such as: are you “able to adapt to change,” “can (you) deal with whatever comes,” to “can handle unpleasant feelings.” This scale has shown good reliability and validity, with Cronbach alpha of 0.88 in 334 cancer patients and 122 breast cancer patients (Matzka et al., 2016; Scali et al., 2012).
The Visual Analog Scale (VAS), a valid and reliable tool, was used to assess the patients’ psychological distress symptoms of anxiety, stress, and depression at pre- and post assessment. It is accurate and sensitive for rating subjective symptoms and has been found to be less burdensome for research participants (Benedetto and Sheehan, 2014; Labaste et al., 2019; Lesage et al., 2012). Three individual visual analog scales of anxiety, stress, and depression were measured on a 0 to 10 scale over the past 7 days (Benedetto and Sheehan, 2014; Labaste et al., 2019; Lesage et al., 2012). The scale ranged from “0” (no anxiety, stress, and depression) to “10” (worst anxiety, stress, and depression).
Functional Assessment of Cancer Therapy – General (FACT-G) is a 27-item questionnaire that measures QOL in four domains of health: physical, functional, social, and emotional wellbeing (Cella et al., 1993). The study used FACT-G to measure patients’ QOL at pre- and post assessment timepoints. Questions range from “I am forced to spend time in bed,” “I feel close to my friends,” “I worry about dying,” to “I am able to enjoy life.” FACT-G is a reliable and psychometric sound instrument with a Cronbach alpha of >0.89 that has been used in a variety of cancer patients (Cella, 2012; Cheng, 2007; Thomas et al., 2004).
Statistical analysis
An a priori power analysis calculation was conducted using G*Power Version 3.1.9.4 with the plan to run a Mixed ANOVA with 2 groups, each measured at 2 timepoints, a medium effect size, alpha = 0.05, and power = 0.80. This power analysis indicated a total sample size of 98 was required. The study aimed to recruit 150 patients from a wide variety of racial and ethnic groups, gender, and over the age of 18. Due to unexpected delays in chemotherapy treatments, resulting in missed appointments and extended study timelines, the final enrolled sample was N = 100 (Control N = 48; Intervention N = 52). This remains a substantially larger sample size than many comparable studies found in the aromatherapy literature. All cases were included in the analysis unless data on the variable of interest were missing.
Descriptive statistics (frequency counts) were used to examine the use of antiemetic medication for the intervention and control groups across multiple visits. Frequency counts were also used to analyze the type of antiemetic medication reported by condition as well as common non-pharmacological interventions used by condition across multiple visits. To assess severity of acute and delayed chemotherapy-induced nausea, median ratings from the MAT were aggregated across all timepoints and compared by condition. To assess severity of acute and delayed chemotherapy-induced vomiting, the proportion of participants who reported vomiting on the MAT was averaged across study timepoints and compared by condition.
To determine the effect of aromatherapy inhalation on resilience and QOL, mixed analysis of variance (ANOVA) was used to determine if change from baseline to post-intervention differed by condition (intervention vs control) on FACT-G Total Score and CD-RISC Total Score. To determine the effect of aromatherapy inhalation on psychological distress and resilience and QOL subscales, the Wilcoxon Signed Rank Test was implemented. Specifically, we examined whether anxiety, stress, or depression changed significantly from baseline to post separately for the intervention group and the control group. This test was also used to determine whether FACT-G and CD-RISC subscales changed significantly from baseline to post for either intervention or control groups. For all significance tests, significance was determined based on p < .05.
Results
Demographic and sample characteristics (pre-assessment demographic checklist) The study included a total of 100 participants randomly assigned to either the control group (48%) or the intervention group (52%). More than 52% of participants in the control group and 67% in the intervention group were female. A majority of participants in both groups identified as Hispanic/Latino(a) and were either married or in a relationship and were unemployed or retired (see Table 1 for additional demographic information).
Effect on chemotherapy-induced nausea and vomiting (MAT)
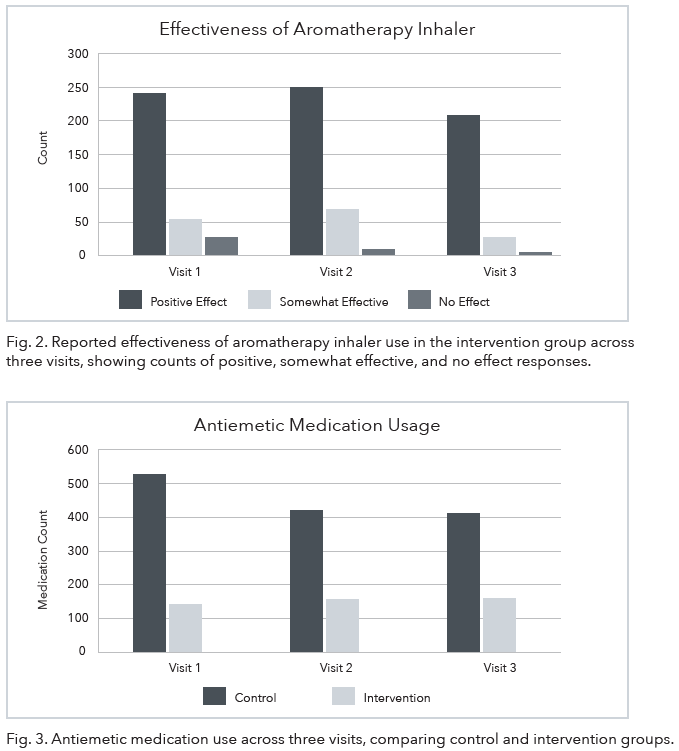
The study hypothesized that aromatherapy inhalation would reduce CINV. The results showed median ratings of severity of acute nausea averaged 5 (SD = 0.58) for the control group and 4 (SD = 0.00) for the intervention group. Median delayed nausea averaged 4.5 (SD = 0.76) for the control group and 4 (SD = 0.00) for the intervention group. The average proportion of control group participants who reported acute vomiting was 11.1 (SD = 2.43) compared to 7.77 (SD = 3.95) in the intervention group. The average proportion of intervention group participants who reported delayed vomiting in the control group was 11.89 (SD = 4.79) compared to 5.95 (SD = 0.00) in the intervention group. In addition, based on the aromatherapy diary, intervention group participants reported positive effects from aromatherapy use (see Fig. 2).
Use of pharmacological and non-pharmacological interventions (Antiemetic Diary)
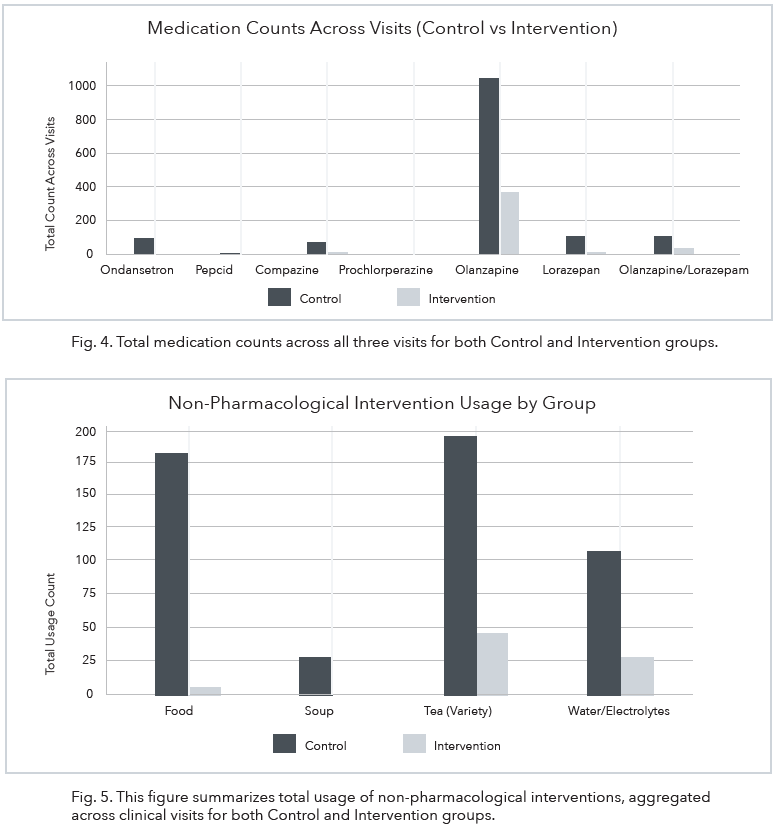
The use of the aromatherapy inhaler resulted in a reduction in antiemetic medication usage between the control group and the intervention group (see Fig. 3). The change in frequency of antiemetic medication use in the control group was 9.89 (SD = 1.41) compared to 3.06 (SD = 0.35) in the intervention group. The control group reported 522 instances of medication usage at visit one; 417 instances of medication usage at visit two; and 409 instances of medication usage at visit three. The intervention group reported 136 instances of medication usage at visit one; 145 instances of medication use at visit two; and 142 instances of medication use at visit three. Ondansetron was predominantly used by both the control and intervention groups. However, other medications including prochlorperazine, olanzapine, and lorazepam were used by both groups (see Fig. 4).
Similar to the patterns observed for pharmacological interventions, the control group also reported higher rates of the use of non-pharmacological interventions including tea, water/ electrolytes, soup, and food compared to the intervention group (see Fig. 5).
Effect on resilience (CD-RISC)
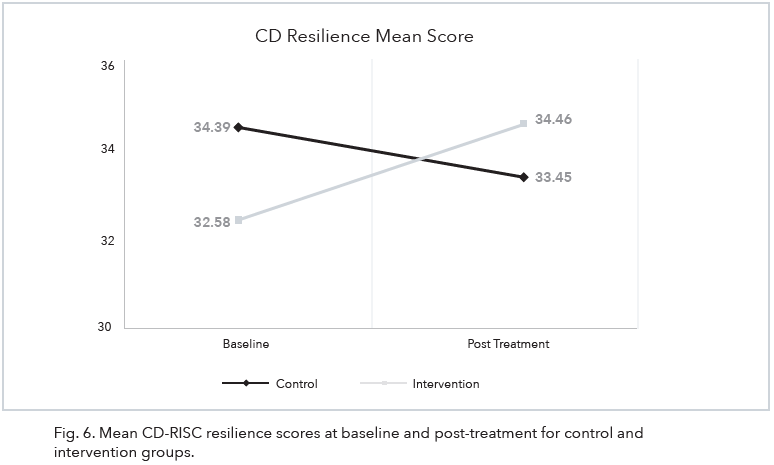
The change from baseline to post-treatment on the CD-RISC was not statistically significant, F (1, 16) = 0.00, p = .986. Moreover, this change did not differ by condition, F (1, 16) = 1.12, p = .307. Examination of individual CD-RISC subscales for the control group did not reveal any significant differences on Flexibility (p = .476), Self-Efficacy (p = .585), Emotion Regulation (p = .808), Optimism (p = .183), and Cognitive Focus (p = .789). For the intervention group, significant changes were observed in Flexibility (p = .036), but not on Self-Efficacy (p = .430), Emotion Regulation (p = .169), Optimism (p = .934), and Cognitive Focus (p = .448). An interesting finding is that CD resilience mean score from baseline to post-intervention increased in the intervention group as compared to a decrease in the control group (see Fig. 6).
Effect on psychological distress (VAS)
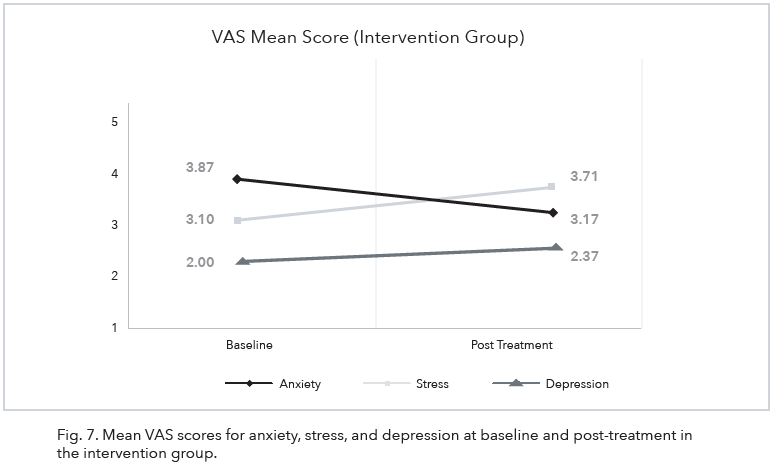
In the control group, there was no significant change as a result of the intervention on anxiety (p = .723), stress (p = .587), or depression (p = .786). Similarly, no significant differences were found among the intervention group on anxiety (p = .798), stress (p = .400), or depression (p = .180). Of note, the intervention group VAS mean score showed a decrease in anxiety (3.87–3.17) from baseline to post-intervention (see Fig. 7).
Effect on quality of life (FACT-G)
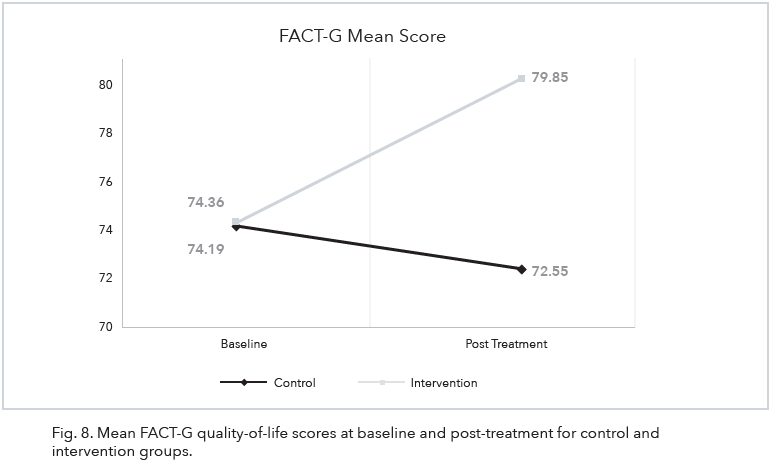
The change from baseline to post-treatment on the FACT-G total score was not statistically significant, F (1, 16) = 0.23, p = .636. Moreover, this change did not differ by condition, F (1, 16) = 0.02, p = .885. Examination of individual FACT-G subscales for the control group found a significant change in Emotional Wellbeing (p = .031) but not Physical Wellbeing (p = .265), Social Wellbeing (p = .415), or Functional Wellbeing (p = .364). For the intervention group, no significant changes were observed on Physical Wellbeing (p = .942), Social Wellbeing (p = .314), Emotional Wellbeing (p = .145), or Functional Wellbeing (p = .661). However, the FACT-G mean score from baseline to post-intervention increased in
the intervention group as compared to a decrease in the control group (see Fig. 8).
DISCUSSION
The findings from this randomized clinical trial highlight the potential benefits of 9 aromatherapy inhalation as a complementary therapy for managing CINV. Based on this study’s results, the use of the aromatherapy inhaler had positive effects on participants. The intervention group used antiemetic medication at a much lower rate, had an increase in resilience, a decrease in anxiety, and a greater sense of wellbeing. These findings suggest that there may be clinically meaningful differences for patients undergoing chemotherapy that experience CINV.
Study implications for practice
Aromatherapy inhalation in congruent with standard antiemetic therapies offers several advantages: (a) cost-effective, (b) useability (e.g., clinical, home, or on-the-go), (c) minimal side effects, and (d) low risk of drug-drug interactions (Hines et al., 2018). Advantages highlight aromatherapy as a complementary therapy for managing CINV while enhancing patient care, comfort, and well-being during chemotherapy (Toniolo et al., 2021). Aromatherapy can be integrated into holistic medical programs offered by hospitals that provide acupuncture and massage therapy services during chemotherapy. Integrative medicine programs have been shown to improve patient outcomes, reduce treatment related symptoms, and promote overall well-being. The improved patient outcomes demonstrate the potential benefits of introducing a dedicated holistic service within outpatient oncology centers, particularly for managing CINV and improving patient experiences. However, aromatherapy has accessibility limitations because of insurance coverage—aromatherapy is not reimbursable—creating barriers for patients who could benefit from this complementary approach (Farrar and Farrar, 2020).
Establishing a holistic service in oncology settings could address a crucial gap in care by providing patients with additional non-pharmacological options to alleviate treatment related symptoms. By incorporating evidence-based complementary therapies like aromatherapy inhalation into routine care, healthcare providers can adopt a more patientcentered approach, prioritizing the physical and emotional well-being of individuals undergoing chemotherapy. Furthermore, implementing such programs aligns with the
growing trend toward integrative care in leading healthcare institutions and offers an opportunity to advance patient care standards in outpatient oncology settings.
This clinical trial is significant because it describes the feasibility and benefits of using aromatherapy as an adjunct to conventional treatment. for CINV. The findings support the idea that low-cost, easily accessible therapies can enhance a patient’s QOL without imposing additional risks or complexity on treatment regimens. Moving forward, integrating holistic services into oncology care can bridge the gap between medical and supportive care, ensuring that patients receive comprehensive and compassionate treatment throughout their cancer journey.
LIMITATIONS
This study had several limitations. First, although larger sample sizes typically result in more reliable and generalizable findings, the sample size had to be reduced from 150 to 100 patients. The sample size of 100 patients remains to be one of the largest nurse-driven clinical trials at the conclusion of this study. Nevertheless, some of the significance tests for the secondary outcomes may have been underpowered. This reduction was a result of delays in chemotherapy treatments because of adverse effects of the chemotherapy treatments, which led to missed appointments and extended the overall study timeline.
Second, the number of study visits could have been reduced from four to three, to help avoid further prolongation of the study and ease the demand for additional nursing staff to conduct these visits. Additionally, the study’s unblinded design is another limitation, as aromatherapy is susceptible to expectancy effects and reporting bias due to the subjective nature of smell (Ahn et al., 2024). Consequently, the observed reduction in antiemetic usage may reflect a strong placebo or psychological effect rather than purely the physiological action of the essential oils. However, the study’s objective was not to isolate the mechanism by comparing aromatherapy against a placebo, a process often compromised by the difficulty of creating an effective sham inhaler (Farrar and Farrar, 2020). Instead, the aim was to assess the total effectiveness and external validity of providing patients with a complementary, patient-controlled tool alongside standard antiemetic care. This design prioritizes determining the therapy’s practical, real-world impact on symptom management and the reduction of pharmacologic medication reliance within a clinical pathway.
Last, the use of handwritten self-report instruments required reminders from the study team members. Converting the instruments to an electronic version may have facilitated the documentation process. To address these limitations, future research should focus on larger sample sizes and long-term studies to evaluate the sustained effects of aromatherapy on CINV.
To accurately measure and compare CINV incidence across studies, researchers must consider the variability in chemotherapy regimens and the subjective nature of CINV. In addition, the FDA classifies aromatherapy as a drug when it is intended for therapeutic purposes, such as disease treatment or prevention, or to influence bodily functions (FDA, 2023). The FDA regulations in Title 21 CFR Part 312 generally state that an experiment in which a drug is administered to human participants is subject to the Investigational New Drug (IND) reporting requirements (FDA, 2025). This study was initially referred to the FDA, prior to IRB approval, due to its therapeutic objectives. Given these therapeutic aims, the
FDA categorized aromatherapy as a drug within the context of our study. Consequently, we would be required to submit an IND application to proceed with the clinical trial. However, since the current study structure did not raise safety concerns and that previous studies successfully employed essential oil aromatherapy to manage CINV in cancer patients, and given the established safety profile of QueaseEASE in pediatric and adult post-surgical populations, the FDA exercised enforcement discretion and no IND reporting requirements were expected for this study. This allowed us to commence the study with a delay of a few months.
CONCLUSION
This study provides preliminary evidence supporting the feasibility and potential benefits of aromatherapy inhalation as a complementary approach for managing CINV for patients receiving moderate to HEC. While the findings suggest that aromatherapy may be a safe, well-tolerated, and accessible adjunct to standard antiemetic therapy, further large-scale randomized controlled trials are needed to confirm its efficacy and generalizability. Given its low cost, ease of use, and minimal reported side effects, aromatherapy represents a promising supportive care option that warrants continued investigation in oncology settings.
CREDIT AUTHORSHIP CONTRIBUTION STATEMENT
Debbie Anglade: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Resources, Project administration, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization.
Caterine Diaz: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Resources, Project administration, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization.
Roberto Roman Laporte: Writing – review & editing, Writing – original draft, Software, Project administration, Formal analysis, Data curation.
Karina Gattamorta: Writing – review & editing, Writing – original draft, Validation, Methodology, Formal analysis, Conceptualization.
Sameena F. Sheikh-Wu: Writing – review & editing, Writing – original draft, Software, Project administration, Methodology, Conceptualization.
Yhenifer Diaz Granados: Writing – original draft, Investigation, Data curation, Conceptualization.
Hoyan Ng-Chen: Writing – original draft, Methodology, Conceptualization.
Junet Alvarez: Writing – original draft, Investigation, Data curation, Conceptualization.
Lisa F. Rosen: Writing – review & editing.
Joseph Pizzolato: Writing – review & editing, Methodology, Investigation, Conceptualization. Declaration of competing interest
Roberto Roman Laporte – Provost at Miami Regional University, Miami Springs, Florida.
Sameena F. Sheikh-Wu – Dr. Sheikh-Wu does not have a current affiliation. At the time of the study, she was affiliated with the School of Nursing & Health Studies, University of Miami, Coral Gables, Florida.
ACKNOWLEDGEMENT
The study team wants to acknowledge Stephanie Carver, BSN, RN, and Soothing Scents, Inc., for the in-kind product donation.
REFERENCES
Adel, N., 2017. Overview of chemotherapy-induced nausea and vomiting and evidencebased therapies. Am. J. Manag. Care 23 (14 Suppl. l), S259–S265. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref1
Ahmad, M., 2016. Tool development to assess nausea and vomiting among patients receiving chemotherapy. Int. J. Cancer. Oncol. 3, 1–5. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref2
Ahn, J.H., Kim, M., Kim, R.W., 2024. Effects of aromatherapy on nausea and vomiting in patients with cancer: a systematic review and meta-analysis of randomized controlled trials. Compl. Ther. Clin. Pract. 55, 101838. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref3
American Cancer Society, 2020. Managing Nausea and Vomiting at Home. American Cancer Society, Atlanta. Retrieved from. https://www.cancer.org/treatment/treat ments-and-sideeffects/physical-side-effects/nausea-and-vomiting/nausea-and- vomiting.html.
Asay, K., Olson, C., Donnelly, J., Perlman, E., 2018. The use of aromatherapy in postoperative nausea and vomiting: a systematic review. J. Perianesth. Nurs. 34 (3), 502–516. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref5
Barbour, S.Y., 2017. Management of patients with chemotherapy-induced nausea and vomiting. J. Adv. Pract. Oncol. 8 (3), 303–308. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref6
Benedetto, M.D., Sheehan, M., 2014. Evaluation of the cardiac depression visual analogue scale in a medical and non-medical sample. Psychol. Health Med. 19 (1), 93–102. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref7
Burns, R., Anstey, K., 2010. The connor–davidson resilience scale (CD-RISC): testing the invariance of a uni-dimensional resilience measure that is independent of positive and negative affect. Pers. Indiv. Differ. 48 (5), 527–531. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref8
Cao, J., Chen, C., Wang, Y., Liu, M., Han, X., Li, H., 2024. A nurse-led multidomain intervention to improve the management of chemotherapy-induced nausea and vomiting in patients with head and neck cancers: a randomized controlled trial. Eur. J. Oncol. Nurs. 70, 102615. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref9
Cella, D., 2012. Assessment methods for quality of life in cancer patients: the FACIT measurement system. Int. J. Pharmaceut. Med. 14, 78–81. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref10
Cella, D.F., Tulsky, D.S., Gray, G., Sarafian, B., Linn, E., Bonomi, A., Brannon, J., 1993. The functional assessment of cancer therapy (FACT) scale: development and validation of the general measure. J. Clin. Oncol. 11 (3), 570–579. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref11
Cheng, K.K., 2007. Oral mucositis and quality of life of Hong Kong Chinese patients with cancer therapy. Eur. J. Oncol. Nurs. 11 (1), 36–42, 2007 Feb. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref12
Dranitsaris, G., Molassiotis, A., Clemons, M., Roeland, E., Schwartzberg, L., Dielenseger, P., Aapro, M., 2017. The development of a prediction tool to identify cancer patients at high risk for chemotherapy-induced nausea and vomiting. Ann. Oncol. 28 (6), 1260–1267. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref13
Duff, K., 2009. Aromatherapy: the power of scents. Townsend Letter 316, 86. Retrieved from.
https://link-gale-com.access.library.miami.edu/apps/doc/A210653401/AO NE?u=miami_
richter&sid=AONE&xid=d8fce3d7.
Escobar, Y., Cajaraville, G., Virizuela, J.A., ´Alvarez, R., Mu˜noz, A., Olariaga, O., et al., 2015. Erratum to: incidence of chemotherapy-induced nausea and vomiting with moderately emetogenic chemotherapy: ADVICE (actual data of vomiting incidence by chemotherapy evaluation) study. Support. Care Cancer 23 (9), 2841–2841. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref15
Farahani, M.A., Afsargharehbagh, R., Marandi, F., Moradi, M., Hashemi, S.-M., Moghadam, M.P., Balouchi, A., 2019. Effect of aromatherapy on cancer complications: a systematic review. Compl. Ther. Med. 47, 102169. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref16
Farrar, A.J., Farrar, F.C., 2020. Clinical aromatherapy. Nurs. Clin. 55 (4), 489–504, 2020 Dec. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref17 FDA, 2023. https://www.fda.gov/cosmetics/cosmetic-products/aromatherapy FDA, 2025. https://www.govinfo.gov/content/pkg/CFR-2025-title21-vol5/pdf/CFR-2025-title21-vol5-part312.pdf
Gupta, K., Walton, R., Kataria, S.P., 2021. Chemotherapy-induced nausea and vomiting: pathogenesis, recommendations, and new trends. Cancer Treat Res Commun 26 (100278),
http://refhub.elsevier.com/S1462-3889(26)00022-0/sref20 Harris, P.A., Taylor, R., Minor, B.L., et al., 2019. The REDCap consortium: building an international community of software platform partners. J. Biomed. Inf. 95, 103208. https://doi.org/10.1016/j.jbi.2019.103208, 2019
Hines, S., Steels, E., Chang, A., Gibbons, K., 2018. Aromatherapy for treatment of postoperative nausea and vomiting. Cochrane Database Syst. Rev. 10 (3) (3): CD007598. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref22
Labaste, F., Ferr´e, F., Combelles, H., Rey, V., Foissac, J., Senechal, A., Minville, V., 2019. Validation of a visual analogue scale for the evaluation of postoperative anxiety: a prospective observational study. Nurs. Open 6 (4), 1323–1330. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref23
Lesage, F.X., Berjot, S., Deschamps, F., 2012. Clinical stress assessment using a visual analogue scale. Occup. Med. (Oxf.) 62 (8), 600–605. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref24
Mapp, C.P., Hostetler, D., Sable, J.F., Parker, C., Gouge, E., Masterson, M., Willis- Styles, M., Fortner, C., Higgins, M., 2020. Peppermint oil: evaluating efficacy on nausea in patients receiving chemotherapy in the ambulatory setting. Clin. J. Oncol. Nurs. 24 (2), 160–164. https://doi.org/10.1188/20.CJON.160-164
Matzka, M., Mayer, H., K¨ock-H´odi, S., Moses-Passini, C., Dubey, C., Jahn, R., Eicher, M., Relationship between resilience, psychological distress and physical activity in cancer patients: a cross-sectional observation study. PLoS One 11 (4). http://refhub.elsevier.com/S1462-3889(26)00022-0/sref27
Molassiotis, A., Coventry, P.A., Stricker, C.T., Clements, C., Eaby, B., Velders, L., Rittenberg, C., Gralla, R.J., 2007a. Validation and psychometric assessment of a short clinical scale to measure chemotherapy-induced nausea and vomiting: the MASCC antiemesis tool. J Pain Symptom Manage. Aug 34 (2), 148–159. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref28
Molassiotis, A., Coventry, P.A., Stricker, C.T., Clements, C., Eaby, B., Velders, L., Gralla, R.J., 2007b. Validation and psychometric assessment of a short clinical scale to measure chemotherapy-induced nausea and vomiting: the MASCC antiemesis tool. J. Pain Symptom Manag. 34 (2), 148–159. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref29
Mosa, A.S.M., Hossain, A.M., Lavoie, B.J., Yoo, I., 2020. Patient-related risk factors for chemotherapy-induced nausea and vomiting: a systematic review. Front. Pharmacol. 11, 329, 2020 Apr 1. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref30
Ng, T.L., Hutton, B., Clemons, M., 2015. Chemotherapy-induced nausea and vomiting: time for more emphasis on nausea? Oncologist 20 (6), 576–583. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref31
Scali, J., Gandubert, C., Ritchie, K., Soulier, M., Ancelin, M.L., Chaudieu, I., 2012. Measuring resilience in adult women using the 10-items connor-davidson resilience scale (CD-RISC). Role of trauma exposure and anxiety disorders. PLoS One 7 (6), e39879, 2012. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref32
Thomas, B.C., Pandey, M., Ramdas, K., Sebastian, P., Nair, M.K., 2004. FACT-G: reliability and validity of the Malayalam translation. Qual. Life Res. 13 (1), 263–269. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref33
Toniolo, J., Delaide, V., Beloni, P., 2021. Effectiveness of inhaled aromatherapy on chemotherapy-induced nausea and vomiting: a systematic review. J. Alternative Compl. Med. 27 (12), 1058–1069, 2021 Dec. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref34
Urbaniak, G.C., Plous, S., 2013. Research Randomizer (Version 3.0). Computer Software. http://www.randomizer.org/.
Yamaguchi, M., Ogawa, T., Watanabe, M., Anami, S., Kamigaki, S., Nishikawa, N., Ono, T., Furukawa, H., 2009. Gan to kagaku ryoho, 36 (10), 1691–1696. http://refhub.elsevier.com/S1462-3889(26)00022-0/sref36
Yokoe, T., Hayashida, T., Nagayama, A., Nakashoji, A., Maeda, H., Seki, T., Kitagawa, Y., 2018. Effectiveness of antiemetic regimens for highly emetogenic chemotherapy- induced nausea and vomiting: a systematic review and network meta-analysis. Oncologist 24 (6), E347–E357.
http://refhub.elsevier.com/S1462-3889(26)00022-0/sref37
